In February 2026, Novo Nordisk’s announcement of Phase 3 clinical data for CagriSema once again sent shockwaves through the global GLP-1 drug market. The data showed that this combination drug outperformed the company’s star product, semaglutide, in terms of weight loss efficacy, dropping another bombshell on the market. However, beyond the spotlight of clinical success, a fundamental challenge looms for all market entrants: the core patent for semaglutide is set to expire in China on March 20, 2026. The massive market gap and the flood of biosimilars are pushing production capacity constraints and cost pressures to their limits.

On one hand, global demand for tirzopentide and semaglutide is growing exponentially; on the other, the complex production processes for these peptides make it difficult to simultaneously achieve high purity, high yield, and low costs. This is not merely a race for production capacity, but a profound contest involving biomanufacturing processes, equipment, and engineering.
-
Purification of Complex Structures: From Reactors to Separation Equipment
The biological activity of peptide drugs, particularly long-chain peptides, is highly dependent on their precise three-dimensional structure. As a result, the manufacturing process requires far stricter control over impurities—especially host cell proteins (HCPs) and residual DNA—than is the case with traditional chemical drugs. Even the slightest sequence error, misfolding, or missing chemical modification can lead to a loss of efficacy or trigger an immune response.
Currently, one of the primary methods for producing GLP-1 peptide drugs is through biological fermentation (recombinant DNA technology), and in this process, the precise control capabilities of bioreactors play a foundational role. They not only serve as an environment that fosters cell growth and product expression but are also key to controlling the impurity profile from the source. Take the HOLVES Eu210 series bioreactors as an example: their advanced control systems can maintain critical parameters such as pH, dissolved oxygen (DO), and temperature within extremely narrow fluctuation ranges, precisely simulating the physiological environment found in the body. By guiding cells toward metabolic pathways that efficiently synthesize the target product while simultaneously suppressing stress pathways that lead to the release of byproducts and human-derived contaminants, the Eu210 series significantly reduces the burden on downstream purification, ensuring that every batch of fermentation broth provides a stable, high-quality starting point.
Currently, one of the primary methods for producing GLP-1 peptide drugs is through biological fermentation (recombinant DNA technology), and in this process, the precise control capabilities of bioreactors play a foundational role. They not only serve as an environment that fosters cell growth and product expression but are also key to controlling the impurity profile from the source. Take the HOLVES Eu210 series bioreactors as an example: their advanced control systems can maintain critical parameters such as pH, dissolved oxygen (DO), and temperature within extremely narrow fluctuation ranges, precisely simulating the physiological environment found in the body. By guiding cells toward metabolic pathways that efficiently synthesize the target product while simultaneously suppressing stress pathways that lead to the release of byproducts and human-derived contaminants, the Eu210 series significantly reduces the burden on downstream purification, ensuring that every batch of fermentation broth provides a stable, high-quality starting point.
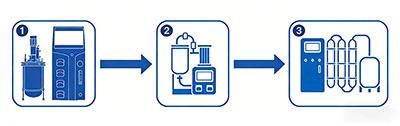
Once the target peptide is secreted into the culture medium or remains intracellularly, the real challenge has only just begun. Culture medium or cell lysates are complex mixtures containing tens of thousands of impurities, such as cell debris, contaminating proteins, nucleic acids, and endotoxins. The goal of separation and purification is akin to “finding a needle in a haystack”—to obtain the target peptide in high purity. This is where tangential flow filtration systems and membrane separation equipment take center stage. TFF systems efficiently perform cell separation, concentration, and medium exchange, enabling highly efficient initial screening. Chromatographic purification, particularly continuous chromatography systems, performs precise fractionation and purification based on the molecular size, charge, hydrophobicity, and other characteristics of different substances.
-
Breakthrough Efficiency
Low production efficiency is the primary factor driving up costs. Traditional batch-based cultivation and purification result in lengthy peptide production cycles, low equipment utilization, and yield losses at each purification step.
How can this be solved? The advent of high-throughput bioreactors has hit the “fast-forward button” for process optimization. As we discussed in our previous article, “High-Throughput Fermentation Unlocks the Potential of Parallel Bioreactors,” by driving multiple micro-reactors in parallel, this technology can screen for optimal engineered strains, medium formulations, and culture conditions at an unprecedented speed. But its value extends far beyond R&D. The optimized process parameters identified through this screening can directly guide production-scale reactors to achieve high-density cell culture, maximizing the peptide production capacity of microorganisms or cells and boosting yield per unit volume at the source.
Today, production methods are shifting from batch processing to continuous, automated operations. This not only significantly shortens production cycles and improves equipment utilization but also reduces operational errors and the risk of contamination caused by human intervention
How can this be solved? The advent of high-throughput bioreactors has hit the “fast-forward button” for process optimization. As we discussed in our previous article, “High-Throughput Fermentation Unlocks the Potential of Parallel Bioreactors,” by driving multiple micro-reactors in parallel, this technology can screen for optimal engineered strains, medium formulations, and culture conditions at an unprecedented speed. But its value extends far beyond R&D. The optimized process parameters identified through this screening can directly guide production-scale reactors to achieve high-density cell culture, maximizing the peptide production capacity of microorganisms or cells and boosting yield per unit volume at the source.
Today, production methods are shifting from batch processing to continuous, automated operations. This not only significantly shortens production cycles and improves equipment utilization but also reduces operational errors and the risk of contamination caused by human intervention
-
Cost Restructuring
Expensive raw materials, lengthy production cycles, and low yields contribute to the high cost of peptide drugs. However, advancements in modern bioprocessing equipment are focused on fundamentally addressing this issue through integrated and intelligent design.
Integrated automation is key to reducing costs. Under traditional production models, materials must be transferred between multiple independent processing units, which not only increases equipment investment and floor space requirements but also leads to repeated cleaning, sterilization, and transfer losses. Integrating multiple processing units into a single automated system not only effectively reduces intermediate steps but also lowers labor costs and minimizes batch-to-batch variations caused by human error.
Membrane separation systems demonstrate their value in material recycling. During the purification of peptides, large volumes of buffer solution and unreacted raw materials are typically discarded after a single use. Membrane separation systems not only precisely separate the target product but also recover and concentrate these valuable materials for reuse, thereby directly reducing material consumption costs per unit of output and yielding particularly significant economic benefits.
In the final stage, the drying and shaping technology for active pharmaceutical ingredients plays a critical role. Whether through spray drying or freeze-drying, purified peptide solutions are instantly dried into powders that are stable and easy to store and transport. This significantly improves the yield of the final product and facilitates subsequent drug formulation development, making it a crucial step in ensuring the successful transition from API to finished dosage form.
Integrated automation is key to reducing costs. Under traditional production models, materials must be transferred between multiple independent processing units, which not only increases equipment investment and floor space requirements but also leads to repeated cleaning, sterilization, and transfer losses. Integrating multiple processing units into a single automated system not only effectively reduces intermediate steps but also lowers labor costs and minimizes batch-to-batch variations caused by human error.
Membrane separation systems demonstrate their value in material recycling. During the purification of peptides, large volumes of buffer solution and unreacted raw materials are typically discarded after a single use. Membrane separation systems not only precisely separate the target product but also recover and concentrate these valuable materials for reuse, thereby directly reducing material consumption costs per unit of output and yielding particularly significant economic benefits.
In the final stage, the drying and shaping technology for active pharmaceutical ingredients plays a critical role. Whether through spray drying or freeze-drying, purified peptide solutions are instantly dried into powders that are stable and easy to store and transport. This significantly improves the yield of the final product and facilitates subsequent drug formulation development, making it a crucial step in ensuring the successful transition from API to finished dosage form.
As a new generation of drugs like CagriSema continue to set new records for efficacy, future competition in the GLP-1 drug market will increasingly shift from molecular innovation itself to the pursuit of efficient large-scale production. Shifts in market demand require systematic breakthroughs in manufacturing processes. From the precise control of cell culture to the efficient separation of target products and the final formulation of active pharmaceutical ingredients, every technological advancement collectively addresses the ultimate challenge: how to make cutting-edge therapies accessible to everyone who needs them at an affordable price. This is not only the mission of equipment suppliers but also the future toward which the entire biotechnology industry is striving.
NEWS:https://www.clinicaltrialsarena.com/news/novo-nordisks-cagrisema-outperforms-ozempic-in-phase-iii-trial/?cf-view
NEWS:https://www.clinicaltrialsarena.com/news/novo-nordisks-cagrisema-outperforms-ozempic-in-phase-iii-trial/?cf-view




