Over the past few decades, the traditional batch production model—involving feeding, waiting, and then production—has virtually dominated the entire industry. As the biomanufacturing sector transitions from large-scale batch production to high-quality, high-efficiency operations, the limitations of batch production have become increasingly apparent. Continuous biomanufacturing, as the key solution to overcoming these industry bottlenecks, is gradually breaking down application barriers and reshaping the development landscape of the biotechnology industry.
-
Why choose continuous manufacturing?
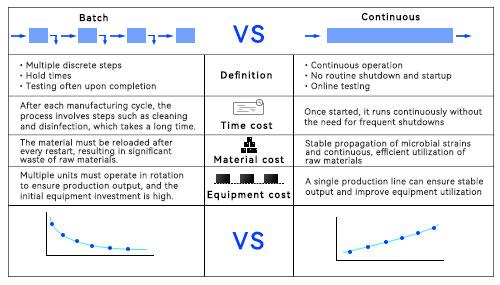
The core issue with batch production lies in the disconnect between unit operations, resulting in the entire process chain being fragmented into several disjointed segments.
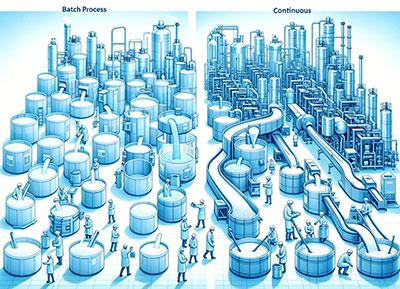
In traditional production, the established process follows a fixed sequence of “feeding—cultivation—harvesting—cleaning—sterilization,” with clear start and stop points at each stage. A significant amount of time is spent on non-productive activities such as sterilization, cleaning, and downtime. Furthermore, downstream processes must wait for upstream batches to be completed before they can begin, often requiring multiple storage tanks for interim holding. This waiting leads to equipment idling, reduced production capacity, and diminished product quality.
More importantly, the very logic of batch scaling is inherently contradictory. Process parameters developed at the laboratory scale cannot be directly scaled up to production scale; they must undergo re-optimization through volume increases of several or even dozens of times, and each such scale-up entails the dual risks of failure and extended timelines. In rapidly evolving fields such as innovative drugs, mRNA, and cell therapy, this approach struggles to meet market demands for small batches, multiple product varieties, and rapid time-to-market, and runs counter to the industry’s goals of green, low-carbon, and highly efficient, intensive production.
More importantly, the very logic of batch scaling is inherently contradictory. Process parameters developed at the laboratory scale cannot be directly scaled up to production scale; they must undergo re-optimization through volume increases of several or even dozens of times, and each such scale-up entails the dual risks of failure and extended timelines. In rapidly evolving fields such as innovative drugs, mRNA, and cell therapy, this approach struggles to meet market demands for small batches, multiple product varieties, and rapid time-to-market, and runs counter to the industry’s goals of green, low-carbon, and highly efficient, intensive production.
-
The Core Benefits of Continuous Manufacturing
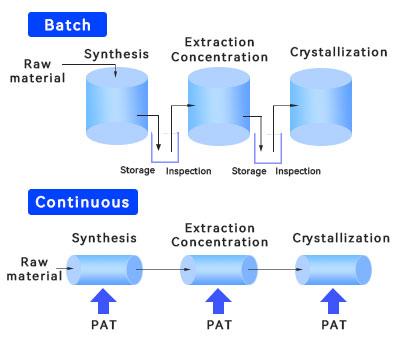
Continuous manufacturing is not a new concept. It has already been successfully implemented in manufacturing sectors such as food production. In the pharmaceutical industry, chemical drugs were the first to adopt continuous manufacturing processes, and the concept of continuous manufacturing has since gradually been adopted in the biopharmaceutical sector. Continuous manufacturing (CM) involves the continuous feeding of input materials and their continuous conversion during the production process, accompanied by the continuous output of finished products. Its core lies in producing intermediates that are immediately transferred to the next process step, thereby creating a highly efficient and seamless assembly-line operation. Numerous research studies have demonstrated that for biopharmaceuticals, promoting continuous production across upstream and downstream processes helps improve product quality, reduce costs, increase efficiency, and enable a more flexible production process.
In terms of efficiency, continuous production enables a significant reduction in equipment footprint and a substantial increase in volumetric productivity. By continuously replenishing culture medium and removing metabolic waste in real time, cells are maintained in a stable growth environment over the long term, and fluctuations in key process parameters are significantly suppressed. Compared to batch production, the continuous production mode involves fewer starts, stops, and transfers throughout the process, reducing the risk of contamination and human interference. This results in higher batch-to-batch consistency, maximizes equipment utilization, and lowers energy and material consumption per unit of product.
In terms of efficiency, continuous production enables a significant reduction in equipment footprint and a substantial increase in volumetric productivity. By continuously replenishing culture medium and removing metabolic waste in real time, cells are maintained in a stable growth environment over the long term, and fluctuations in key process parameters are significantly suppressed. Compared to batch production, the continuous production mode involves fewer starts, stops, and transfers throughout the process, reducing the risk of contamination and human interference. This results in higher batch-to-batch consistency, maximizes equipment utilization, and lowers energy and material consumption per unit of product.
In terms of flexibility, continuous manufacturing offers production capacity flexibility that batch production cannot match. In batch production, capacity is determined by reactor volume, and scaling up requires the construction of larger vessels. In contrast, the modular architecture of continuous production allows production lines to be flexibly configured based on product type and capacity requirements, enabling faster product changeovers. This approach accommodates small-scale pilot testing, pilot-scale production, and full-scale manufacturing, making it better suited to the rapid commercialization of innovative biopharmaceuticals and the need for flexible capacity expansion.
In terms of quality, the fully enclosed production process of continuous manufacturing ensures consistent quality. Leveraging online sensors, real-time analysis, and intelligent control, a data-driven monitoring and control system is established. Because materials flow continuously throughout the entire process, Process Analytical Technology (PAT) can collect quality data in real time at every stage, minimizing process fluctuations and supporting the stable scale-up of the process.
In terms of quality, the fully enclosed production process of continuous manufacturing ensures consistent quality. Leveraging online sensors, real-time analysis, and intelligent control, a data-driven monitoring and control system is established. Because materials flow continuously throughout the entire process, Process Analytical Technology (PAT) can collect quality data in real time at every stage, minimizing process fluctuations and supporting the stable scale-up of the process.
-
Challenges of Continuous Manufacturing
Continuous manufacturing is not merely a simple series of interconnected devices, but rather a highly systematic engineering process that spans three levels: upstream, downstream, and system integration.
Continuous upstream production centers on perfusion culture. Relying on high-performance bioreactors and cell retention devices, it enables stable operation at high cell densities over extended periods, placing high demands on the control precision of the bioreactors and the cell retention system. Minimizing membrane fouling, controlling shear stress, and maintaining a balanced nutrient supply are all critical to continuous culture.
Continuous upstream production centers on perfusion culture. Relying on high-performance bioreactors and cell retention devices, it enables stable operation at high cell densities over extended periods, placing high demands on the control precision of the bioreactors and the cell retention system. Minimizing membrane fouling, controlling shear stress, and maintaining a balanced nutrient supply are all critical to continuous culture.

While upstream perfusion culture has reached a relatively mature stage, the downstream purification unit has long remained the last bastion of batch processing. Continuous downstream purification relies on tangential flow ultrafiltration systems for concentration and medium exchange, continuous chromatography systems for high-efficiency purification, and in-line virus inactivation modules to ensure biosafety. However, mature, standardized protocols for subsequent steps—such as continuous virus inactivation and continuous ultrafiltration—are still lacking. Fluctuations in upstream parameters are directly transmitted to the downstream process. Achieving flow rate matching, parameter buffering, and long-term stability of media represents the core challenges in downstream continuous processing.
To meet the engineering requirements of continuous biomanufacturing, HOLVES has leveraged its years of experience in the R&D and implementation of bioprocessing equipment to develop a comprehensive support solution spanning from process design to operational deployment. We not only provide a full suite of core equipment tailored for continuous production—including upstream bioreactors compatible with perfusion processes that enable high-precision multi-parameter control and long-term aseptic operation, as well as downstream continuous purification equipment covering continuous chromatography, continuous virus inactivation, tangential flow ultrafiltration, and online formulation—but also offer comprehensive support for CIP/SIP, enabling seamless integration between upstream and downstream processes. We customize exclusive continuous processes for clients and provide process validation, GMP compliance guidance, personnel training, and full lifecycle operation and maintenance services. This comprehensive approach lowers the barriers to continuous manufacturing transformation and mitigates operational risks, helping clients efficiently and smoothly transition from batch production to continuous biomanufacturing.
To meet the engineering requirements of continuous biomanufacturing, HOLVES has leveraged its years of experience in the R&D and implementation of bioprocessing equipment to develop a comprehensive support solution spanning from process design to operational deployment. We not only provide a full suite of core equipment tailored for continuous production—including upstream bioreactors compatible with perfusion processes that enable high-precision multi-parameter control and long-term aseptic operation, as well as downstream continuous purification equipment covering continuous chromatography, continuous virus inactivation, tangential flow ultrafiltration, and online formulation—but also offer comprehensive support for CIP/SIP, enabling seamless integration between upstream and downstream processes. We customize exclusive continuous processes for clients and provide process validation, GMP compliance guidance, personnel training, and full lifecycle operation and maintenance services. This comprehensive approach lowers the barriers to continuous manufacturing transformation and mitigates operational risks, helping clients efficiently and smoothly transition from batch production to continuous biomanufacturing.


